Restylane® Facts Part 1
I have been asked a number of questions from my patients about Restylane® and wanted to share with you some important facts about this injectable filler range.
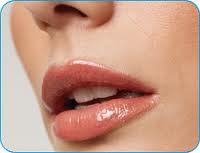
Restylane®is the trade name for a range of injectable fillers with a specific formulation of non-animal sourced hyaluronic acid. Restylane was the first stabilized hyaluronic acid filler on the market and reportedly has been used in over 11 million treatments worldwide. This number of worldwide treatments is quite impressive and speaks for the safety of the product.
In the United States of America, Restylane® was the first hyaluronic acid filler to be approved by the United States Food and Drug Administration (US FDA) for cosmetic injection into sub dermal facial tissues.
As part of our July promotion, we are still offering 10% off Restylane® treatments.
*Please email Jenny@Casas.md or Amy@Casas.md for additional details and to schedule an appointment! And mention you saw this website promotion. We’d love to help you with your goals and answer any questions you may have! Some exclusions apply.
How can I get more information?
Call now for your complimentary consultation.
You may reach us at the Chicago North Shore suburbs of Glenview (847-657-6884).
Dr. Laurie A. Casas and her team will work with you to develop a treatment plan for your cosmetic and aesthetic wishes.
Author: Dr. Laurie A. Casas, www.casas.md – MD, FACS
www.casas.md today!
Submit your own plastic surgery question!
2050 Pfingsten Road – Suite 270 – Glenview, IL 60026
The University of Chicago Medical Center – 5841 S. Maryland Avenue – Chicago, IL 60637
Central Phone: (847) 657-6884