KYBELLA™- Chin Contouring (deoxycholc acid) Injection Chicago
Jump To
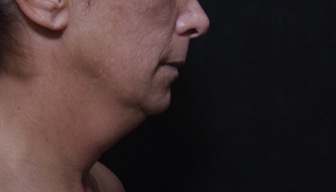
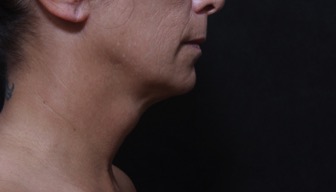
Photo Gallery
View before-and-after pictures of real patients of Dr. Laurie A. Casas
Overview

NEW FDA Approved Non-Surgical Chin and Jawline Contouring Treatment for Men and Women
KYBELLA™(deoxycholic acid)- Chin Contouring Injection to recontour the jawline is the first FDA approved and clinically proven, customizable non surgical treatment that corrects submental fullness caused by moderate to severe excess fat under the chin. This excess fat creates an ill-defined jawline. Over 80% of patients with submental fullness report dissatisfaction with the lack of chin contour. Dr. Laurie Casas, a female ABMS certified Plastic surgeon, a recognized key opinion leader and a national and international educator, has extensive experience with evaluating and treating the aging chin and neck. Dr. Casas is one of a few Plastic Surgeons in the USA chosen by Kythera, the maker of KYBELLA™- Chin Contouring, to train qualified health care professionals in the safe use of this cytolytic drug to decrease submental fullness creating a contoured jawline.Reasons to Use KYBELLA™- Chin Contouring to Reduce Moderate to Severe Submental Fat
Kybella™- Chin Contouring, a deoxycholic acid injection, is a prescription medicine, and when injected provides the first FDA approved and clinically proven non-surgical office procedure that can be customized to correct a double chin. Many patients of all ages are dissatisfied with the appearance of their chin and jawline due to moderate to severe excess fat under their chin, which is also called, submental fat. This creates a fullness and lack of definition along and under the jawline. For over 25 years, Dr. Laurie Casas has offered patients several surgical options to recontour the jawline of patients that want to improve the appearance and profile of their chin and jawline. She has successfully treated submental fullness and the aging neck either with minimally invasive liposuction with or without laser or when necessary with traditional invasive neck lift surgery. These procedures have been the gold standard surgical options, which she will continue to offer patients who require surgery, to recontour their chin and aging neck. Now that KYBELLA™- Chin Contouring is available, Dr. Casas will be able to help patients understand if this new customizable option which dissolves fat through a series of injections may be a better option for their specific chin and jawline contour abnormality. The perfect candidate for non-surgical chin contouring with Kybella- Chin Contouring Injections has well-defined moderate to severe submental fat and has moderate to excellent skin elasticity. A comprehensive consultation will be necessary to evaluate your individual chin and jawline concerns. As always, Dr. Casas will review the pros and cons of all the non-invasive, minimally invasive and traditional procedures that will best treat your chin, jawline and neck.Clinical Results with KYBELLA™- Chin Contouring (deoxycholic acid) Injection
The FDA reviewed two pivotal phase III randomized, double blind, multicenter, placebo controlled clinical trials that evaluated the safety, effectiveness and patient satisfaction of KYBELLA™- Chin Contouring which was used to improve the appearance of chin and jawline in men (15%) and women (85%) between the ages of 19-65, who were dissatisfied with the excess fat in the submental area. Many men and women in the trials stated they experienced visible results after 2-4 treatment sessions that were spaced 4 weeks apart. 79% of the patients, in the combined group of men and women treated with KYBELLA™- Chin Contouring, reported they were satisfied with the appearance of their face and chin. The adverse reactions to the injections with KYBELLA™- Chin Contouring were reported as mild (81%), moderate (17.6%) and severe (1.6%). The most common adverse reactions were reported at the injection site: edema/swelling, hematoma/bruising, pain, numbness, erythema, and induration. O.9% of the adverse reactions had resolved by the end of the study. Proper injection technique and proper injection placement, by a well-trained health care professional, can avoid injury to the marginal mandibular nerve and salivary glands, lymph nodes and muscles. It was found that KYBELLA™- Chin Contouring should not be injected into or in close proximity (1-1.5cm) to salivary glands, lymph nodes, muscles and the marginal mandibular nerve.What to Expect
In the consultation, Dr. Laurie Casas will review your individual case, your expectations and discuss your non-surgical options with KYBELLA™- Chin Contouring . She will also discuss all your minimally invasive and surgical options. She will instruct you with regard to pre treatment preparation, immediate post treatment care , when to return for treatment assessment and when to discuss the need for subsequent treatments. Dr. Casas will explain to you what to expect immediately and after 4-6 weeks. At each assessment 4-6 weeks after treatment she will explain whether further treatments are needed to achieve the result you are expecting. The treatments are performed in the office using local anesthesia to decrease the discomfort from the cytolytic injections with KYBELLA™- Chin Contouring. You will have thorough follow-up, including photo documentation of the result of each treatment sessions at 4-6 weeks post treatment. Dr. Casas is always available should you have questions after your treatment. See more about Dr. Laurie Casas’ KYBELLA™- chin contouring treatment here. Once selected as an appropriate candidate and after full informed consent, Dr. Laurie Casas will use Kybella- Chin Contouring to redefine and recontour your jawline. Kybella- Chin Contouring (deoxycholic acid) Injection is the first FDA approved non-surgical office treatment that reduces moderate to severe submental fullness after a series of 2-6 injection sessions. Dr. Laurie Casas, a female ABMS certified Plastic surgeon and recognized key opinion leader, has been chosen to train qualified physicians on the safe use injectable Kybella- Chin Contouring, a unique cytolytic drug, that dissolves fat under the chin to create an improved contour. In an in depth consultation, Dr. Casas will evaluate your chin, submental and jawline to help you better understand if treatments with injectable Kybella- Chin Contouring will help to redefine your jawline. Here at Casas Aesthetic Plastic Surgery, we also recommend using Alastin TransFORM Body Treatment twice daily after your Kybella procedure. This new technology helps accelerate the body’s natural removal process of post procedure fat debris along with tightening and improving the quality of the skin. You use Alastin TransFORM Body Treatment by applying the product in the area of treatment in the morning and evening.Kybella- Chin Contouring Testimonials
“Kybella- Chin Contouring Got Rid of My Double Chin at 29 and Ulthera tightened the Skin Under My Chin I am a 29 year old female and I was very bothered by my double chin. Regardless of how much I dieted, exercised or lost weight I could always see my double chin in the mirror. I went to Dr. Casas and she suggested the new treatment, Kybella- Chin Contouring. She explained that Kybella- Chin Contouring is a cytolytic agent that dissolves the fat under the chin. I was so thrilled to have an easy, non-surgical solution. She also explained it if there was a little loose skin left after the fat was gone, she could then tighten it with Ulthera. Dr. Casas injected lidocaine in the area first. Then, she injected the Kybella- Chin Contouring which was totally painless because I was numb. I was swollen for a few days. After about 6 weeks, I saw a huge improvement in the area! I was so happy to finally get rid of my ugly double chin. We both noticed because I do not have a deep chin, Ulthera could help tighten the chin under my chin. I had one small treatment virtually painless treatment with Ulthera. After two to three months , and both non surgical treatments, I now have a beautiful neck line. I would recommend Dr. Casas , Kybella- Chin Contouring and Ulthera for anyone bothered by fullness under their chin.” * Individual results may vary
References
- Dayan SH, Jones DH, Carruthers J, et al. A Pooled Analysis of the Safety and Efficacy Results of the Multicenter, Double-Blind, Randomized, Placebo-Controlled Phase 3 REFINE-1 and REFINE-2 Trials of ATX-101, a Submental Contouring Injectable Drug for the Reduction of Submental Fat. Plastic and Reconstructive Surgery 2014;134(4S-1):123 http://journals.lww.com/plasreconsurg/Citation/2014/10001/Prospective,_Multi_Center,_Pivotal_Trial.179.aspx
- James McDiarmid, Jesus Benito Ruiz, Daniel Lee, Susanne Lippert, Claudia Hartisch & Blanka Havlickova Results from a Pooled Analysis of Two bEuropean, Randomized, Placebo- Controlled, Phase 3 Studies of ATX-101 for the Pharmacologic Reduction of Excess Submental Fat
- Ascher B1, Hoffmann K, Walker P, Lippert S, Wollina U, Havlickova B.Efficacy, patient-reported outcomes and safety profile of ATX-101 (deoxycholic acid), an injectable drug for the reduction of unwanted submental fat: results from a phase III, randomized, placebo-controlled study. J Eur Acad Dermatol Venereol. 2014 Dec;28(12):1707-15. http://www.ncbi.nlm.nih.gov/pubmed/24605812
- Rotunda AM, Weiss SR, Rivkin LS. Randomized double-blind clinical trial of subcutaneously injected deoxycholate versus a phosphatidylcholine-deoxycholate combination for the reduction of submental fat. Dermatol Surg. 2009 May;35(5):792-803. doi: 10.1111/j.1524-4725.2009.01130.x. Epub 2009 Apr 6. http://www.ncbi.nlm.nih.gov/pubmed/19397673